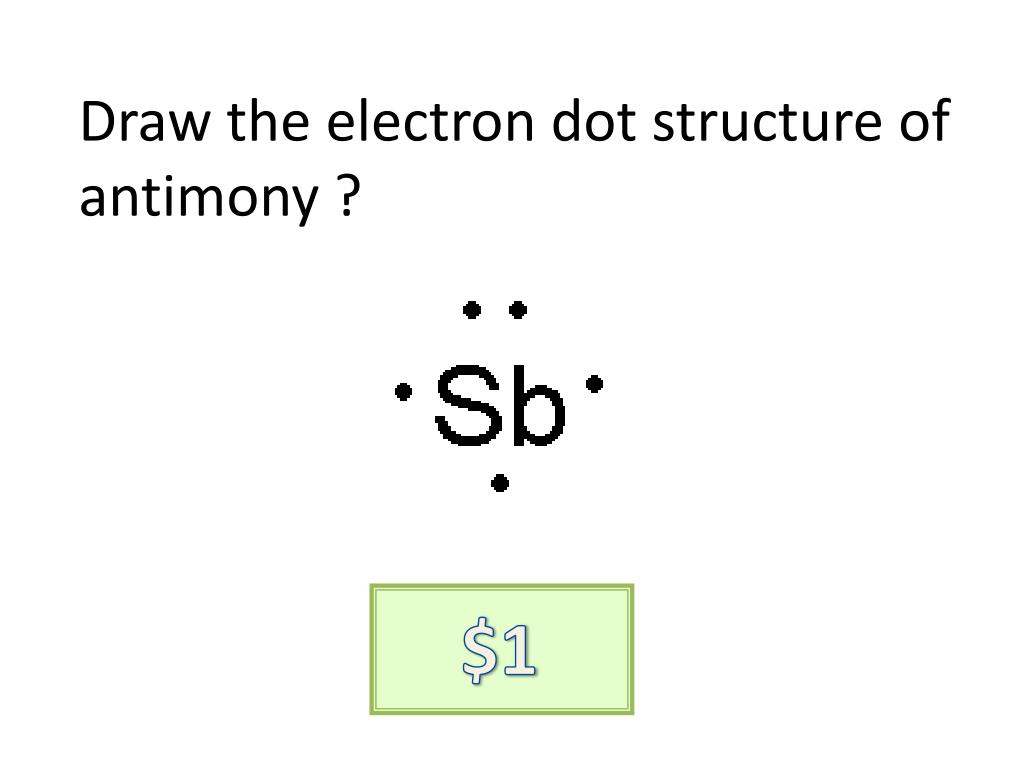
Antimony is the element of nitrogen family hence it has the position below phosphorus. In the solid state structure, the Sb-C bond distances between Sb and. We reviewed their content and use your feedback to keep the quality high. The precise valency of Antimony is 5 since it contains 5 valence electrons in its outer shell. Among pnictogen group Lewis acidic compounds, unusual lewis acidity of Lewis acidic antimony. Who are the experts Experts are tested by Chegg as specialists in their subject area. The hemoglobin molecule contains a similar ring structure, which an iron ion. You can easily understand the number and bonding of Antimony’s valence electrons in the diagram. Antimony and bismuth can lose either their outermost p electrons to form 3+. It’s further useful in depicting the chemical bonding of valence electrons. The diagram explains the interaction of valence electrons of atoms. 
Take the look of Antimony valence electrons with our Lewis dot diagram. The element increases the hardness potency of alloys to make it stronger. Further, the rest of Antimony is useful to use as the pure metal element with the alloys. phase diagrams with the solutes silver, indium, and antimony has been made. Nearly 60% of Antimony is used in the production of flame retardant appliances. The country produces and exports the major part of Antimony to the world.Īntimony has high flame retardation properties hence it’s useful to use with such appliances. China is currently the largest producer of Antimony in the world. The powder form is mainly useful in the production of several medicines in the pharmaceutical domain. 181 Full optical link electrical eye diagram, 412, 413 Full width at half maximum (FWHM), 271, 346 G Gallium antimony, 277 atom position, 276 densities. So, being the Metalloid element Antimony is available both in the powder and metal form.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
